 Carbon dioxide is also formed when fires release carbon stored in vegetation or when we burn wood or fossil fuels. The formation and subsequent ionization of sulfuric acid are shown here:Ĭarbon dioxide is naturally present in the atmosphere because we and most other organisms produce it as a waste product of metabolism. Normal rainwater has a pH between 5 and 6 due to the presence of dissolved CO 2 which forms carbonic acid:Īcid rain is rainwater that has a pH of less than 5, due to a variety of nonmetal oxides, including CO2, SO2, SO3, NO, and NO2 being dissolved in the water and reacting with it to form not only carbonic acid, but sulfuric acid and nitric acid. Table 1. Summary of Relations for Acidic, Basic and Neutral Solutionsįigure 1 shows the relationships between, , pH, and pOH, and gives values for these properties at standard temperatures for some common substances. Unless otherwise noted, references to pH values are presumed to be those at standard temperature (25 ☌) (Table 1). This distinction can be important when studying certain processes that occur at nonstandard temperatures, such as enzyme reactions in warm-blooded organisms. \text) = 6.31Īt this temperature, then, neutral solutions exhibit pH = pOH = 6.31, acidic solutions exhibit pH less than 6.31 and pOH greater than 6.31, whereas basic solutions exhibit pH greater than 6.31 and pOH less than 6.31. The pH of a solution is therefore defined as shown here, where is the molar concentration of hydronium ion in the solution: One such scale that is very popular for chemical concentrations and equilibrium constants is based on the p-function, defined as shown where “X” is the quantity of interest and “log” is the base-10 logarithm: A solution is neutral if it contains equal concentrations of hydronium and hydroxide ions acidic if it contains a greater concentration of hydronium ions than hydroxide ions and basic if it contains a lesser concentration of hydronium ions than hydroxide ions.Ī common means of expressing quantities, the values of which may span many orders of magnitude, is to use a logarithmic scale. The concentrations of these ions in a solution are often critical determinants of the solution’s properties and the chemical behaviors of its other solutes, and specific vocabulary has been developed to describe these concentrations in relative terms. Perform calculations relating pH and pOHĪs discussed earlier, hydronium and hydroxide ions are present both in pure water and in all aqueous solutions, and their concentrations are inversely proportional as determined by the ion product of water ( K w).Express hydronium and hydroxide ion concentrations on the pH and pOH scales. 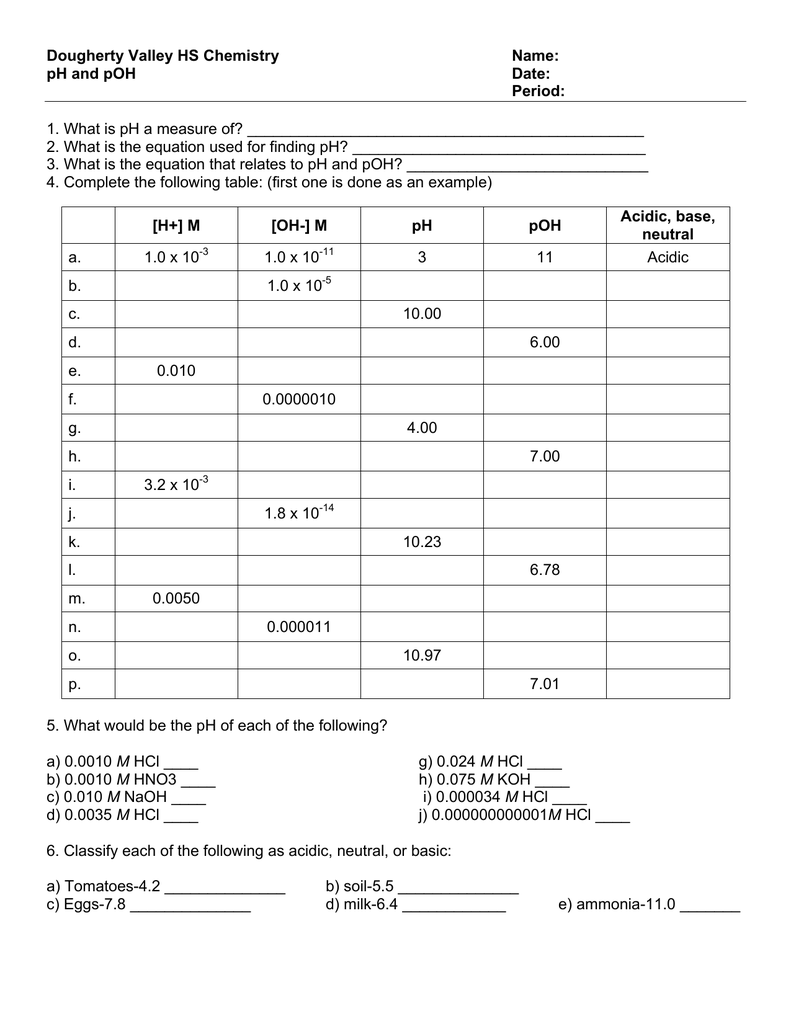 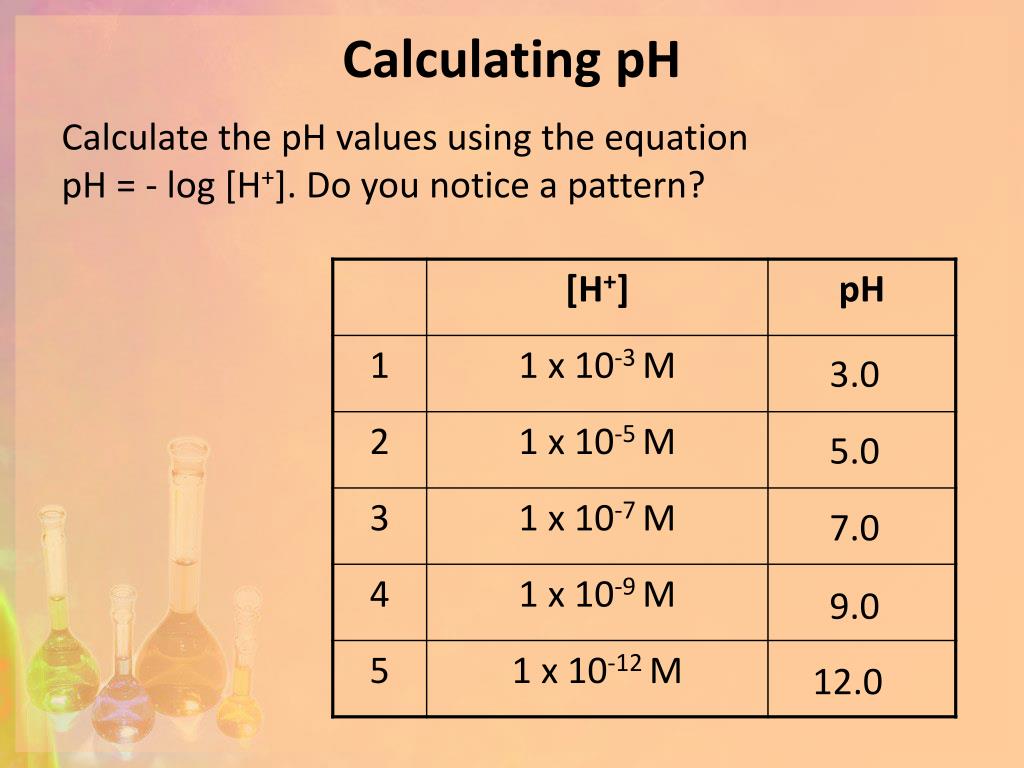
Explain the characterization of aqueous solutions as acidic, basic, or neutral. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
